Healthesystems has previously reported on the growing presence of private-label topical analgesics in workers’ compensation (Topical Analgesics: Expensive and Avoidable, Fall 2013). Payers are beginning to take notice of this continuing trend, and with good reason. From a clinical perspective, private-label topicals offer no greater benefit to the patient than over-the-counter (OTC) alternatives found in retail stores. Like their OTC counterparts, private-label analgesics have not undergone controlled studies to support their clinical efficacy or safety and have not been approved by the U.S. Food and Drug Administration (FDA). Yet in most cases they are exponentially — and unjustifiably — more expensive. So why are these products being prescribed at increasing rates?

In some cases, the perceived difference between expensive private-label topicals and OTC products can encourage prescribing. Research shows that physicians will recommend an OTC medication before a prescription product, as long as they believe both products to have comparable efficacy.1 However, clinical-sounding brand names associated with private-label topicals, such as Medi-Derm, Medrox® Rx and Terocin®, rather than consumer-friendly names such as IcyHot® or BenGay®, infer clinical legitimacy, though these products have not been proven in controlled trials. Some of these products are marketed by manufacturers as having unique formulations and special ingredient blends, when in reality the active ingredients in these products commonly overlap with inexpensive and widely accessible OTC alternatives. Independent wholesalers also use marketing to encourage physician prescribing of these products. As a result, prescribers may believe they are recommending a superior product, without recognizing the safety or cost implications.
The website for a popular physician-dispensing company, which assists prescribers with implementing and conducting in-office dispensing of medications, markets Medrox® pain lotion as a “fantastic alternative for patients seeking narcotics.”2 While it is true that responsible pain management includes the usage of opioid therapy alternatives, when assessing these alternatives, physicians must first consider patient safety and evidence-based guidelines. Prescribing of private-label topical analgesics is not supported by evidence-based guidelines. Furthermore, the FDA has previously issued safety communications regarding the occurrence of serious skin burns in products containing high concentrations of menthol
and methyl salicylate (concentrations greater than 3% and 10%, respectively).3 Of the 22 private-label topical analgesics Healthesystems has observed being prescribed for injured workers, 19 contain menthol and/or methyl salicylate, and 74 percent contain these ingredients at concentrations twice or even three times higher than the recommended FDA thresholds.4 In comparison, there are some OTC alternatives available that contain lower doses of these potentially harmful skin irritants (see Figure below).
The Official Disability Guidelines (ODG) include recommendations for OTC alternatives of these pricey topical analgesics, although it should be noted that the guidelines offer additional direction regarding treatment with specific analgesic ingredients. For example, ODG recommends only the lidocaine 5% patch (Lidoderm® or generics) for patients experiencing neuropathic pain. Any other commercially available forms of lidocaine (e.g., lidocaine 3% cream) are suggested for general numbing or relief of itching. Also of note, capsaicin is deemed appropriate only for patients who have not responded to or are intolerant of other treatments.5 Therefore, topical capsaicin products should not be prescribed as first-line therapy, regardless of cost.
While evidence-based guidelines should be the primary basis of prescribing decisions, in an environment where a large component of outcomes measurement relies on patient-reported pain ratings,6,7 physician perception of patient satisfaction does come into play. Prescribers seeking patient satisfaction with treatment should feel confident in recommending consumer OTC brands, as research demonstrates that the majority of patients trust OTC medications. In a survey conducted in 1,000 U.S. consumers aged 18 or older, OTC medications were the first choice of most respondents to treat minor injury or ailment, and 9 out of 10 respondents believed that OTC medicines are an important part of their family’s overall healthcare.1 This trust is not without reason. OTC products available to consumers directly through their retail store, such as BenGay®, IcyHot® and Aspercreme®, are often produced by major pharmaceutical companies with long-standing FDA relationships.
It is important for prescribers to recognize that patient satisfaction with treatment often correlates with the level of understanding the injured worker has regarding their injury and its treatment. When prescribing OTC topical analgesics, physicians should clearly define the role of these medications in pain management — in the majority of cases, for the temporary relief of minor musculoskeletal pain.
Physician prescribing of private-label topical analgesics has doubled since 2012.4 Based on observed trends, these products are being seen disproportionately within physician-dispensed medications. They are also being dispensed through smaller, independent dispensaries, such as pharmacies associated with the prescribing physician’s practice or “Mom and Pop” shops.
Based on these data, one may infer that the rising trend of private-label topicals is partially incentivized by financial opportunity. State reforms in recent years have sought to reduce costs associated with physician dispensing by limiting reimbursement rates on select drugs (see Physician Dispensing: New Challenges in an Ongoing Battle).8 Under these new regulations, private-label topicals may offer an attractive opportunity for profit, due to their high average wholesale prices (AWPs), which are often disproportional to their cost of manufacture.
Regardless of drivers for increased prescribing, or which stakeholder is dispensing, the injured worker patient leaves the office or pharmacy believing they’ve received a superior medication. In reality, an OTC product from a retail store can offer them comparable efficacy at a much lower cost to the payer.
With prescriptions of private-label topical analgesics continuing to trend upwards, there is a greater need for oversight of these products on the part of the payer and pharmacy benefit manager (PBM). Certain private-label products continue to appear on the list of medications most prescribed. A PBM can work collaboratively with a payer to define appropriate parameters that can best guide decision-making for the claims professional. These parameters should include factors such as patient safety concerns, as well as the fact that evidence-based guidelines do not recommend use of these products. A PBM can also recommended appropriate and more cost-effective OTC alternatives. From a claims professional standpoint, additional education is required regarding private-label topicals, including a familiarity with product names and an understanding regarding the safety and cost implications of these products. Claims professional education is essential to the overall strategy for limiting the use of inappropriate therapies to ensure quality care, while reducing costs associated with these products.
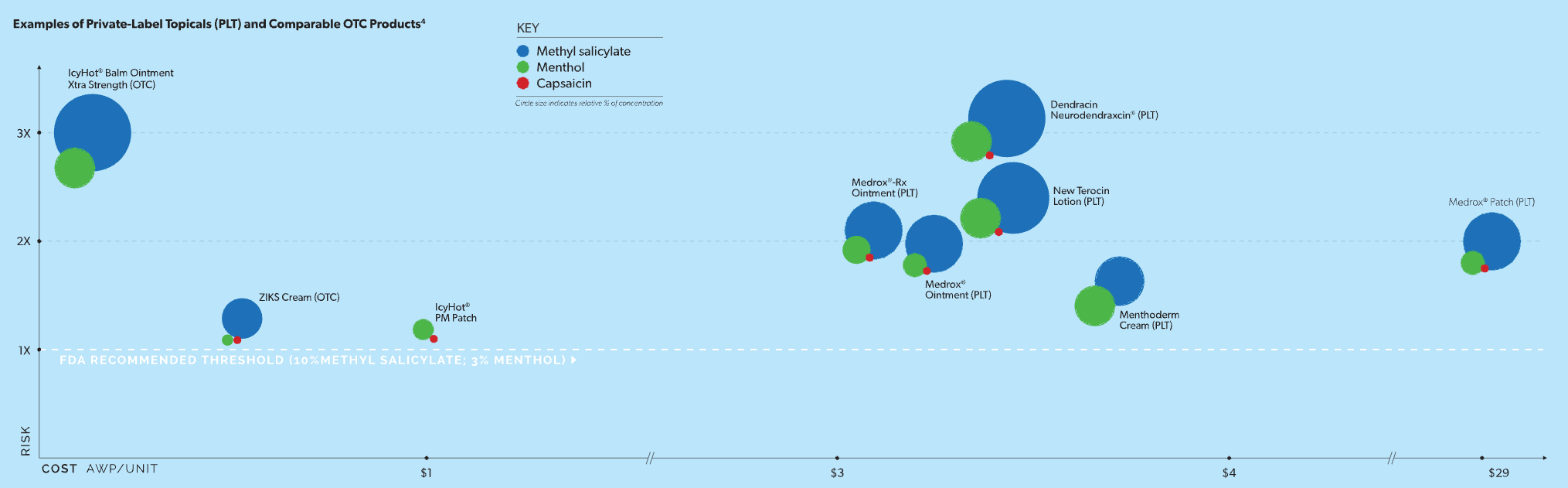
The figure above plots risk vs cost for some examples of private-label analgesics observed by Healthesystems within injured workers’ claims and suitable OTC alternatives.
In addition to the examples above, Healthesystems has collected data and developed recommendations on the following private-label topical analgesics: Aleveer Patch, Biotherm lotion, eLenzaPatch®, Keratek™ gel, LenzaGel®, LenzaPatch®, LidoPro® Ointment, Lidoprofen, Medi-Derm Cream, Terocin Lotion, Terocin Patch, Tru-micin® Lotion, Ultracin Lotion.
While the difference in AWP/unit may only be a couple of dollars, the overall cost adds up. Consider the cost incurred for a prescription of these two very similar products:

Payers may take specific actions to help curb prescribing, including limiting these products from their formularies, or requiring a Letter of Medical Necessity (LOMN) from prescribers to demonstrate a particular patient’s medical need. A pharmacy benefits manager can help define criteria based on appropriateness of therapy, patient safety and cost, as well as provide appropriate alternatives to ensure injured workers are receiving cost-effective therapy that addresses their pain management needs.